Institutional Review Board/ethics Committee What Is the Role of
How do Institutional Review Boards (IRB) and Ethics Committees (EC) touch clinical trials?
- March 16, 2021
- 1:32 pm

Suzanne H.
Sales and Marketing
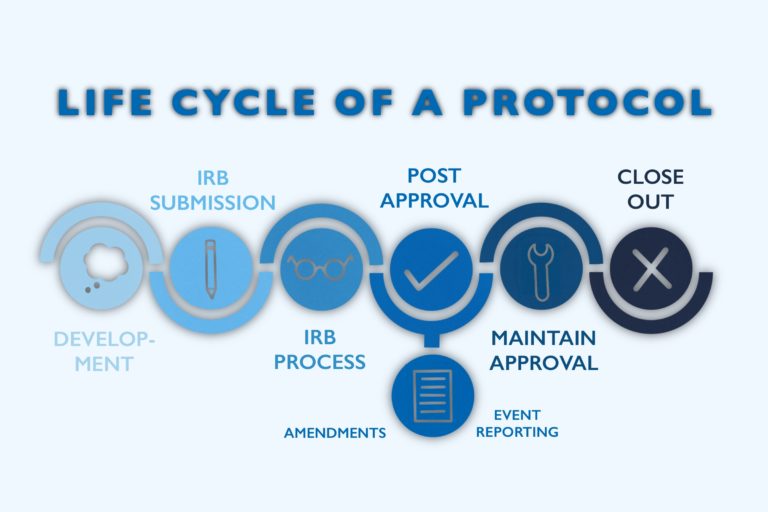
Developing a patient-axial and compliant study is a top priority for any clinical team. Agreement the importance of Institutional Review Boards (IRB) and Independent Ideals Committees (IEC) and their role is critical to a successful clinical trial. Before a trial tin can brainstorm, nevertheless, each aspect of the report must commencement exist reviewed and approved past a designated regulatory body. IRBs and IECs check each component of the clinical trial, from protocol evolution to patient reimbursement and travel back up. Below, y'all will learn about several types of these boards and how Clincierge helps sponsors and CROs continue logistics back up and reimbursement compliant to avoid any study delays.
IRBs and ECs: Oft Asked Questions
What is an Independent Ethics Committee (IEC)?
An Independent Ethics Committee comprises various pharmaceutical industry stakeholders, including doctors, nurses, social workers, chaplains, and community members. These members come up from diverse backgrounds and are stakeholders in the delivery of healthcare. They join together to hash out the clinical trial'south scientific, ethical, and legal ramifications and evaluate the benefit/risk factors involved.
What is the purpose of an Contained Ethics Committee (IEC)?
Ethics Committees ensure that homo subject area research and medical experiments operate according to all national and international laws.
What is an Institutional Review Board (IRB)?
An Institutional Review Board is a formally designated grouping assigned to monitor and review whatever medical research involving man subjects. This squad has the dominance to approve or disapprove research and ask for research modifications to lead to blessing. IRBs operate under United States Food and Drug Assistants (FDA) regulations.
What is the purpose of an Institutional Review Board (IRB)?
Institutional Review Boards are responsible for protecting and assuring that human research subjects' rights and welfare both before they brainstorm their participation (with appropriate protocols in place) and throughout the clinical trial (through periodic study reviews).
What is the difference betwixt an IRB and IEC?
Clinical trials conducted in the European Union are held accountable by Contained Ethics Committees (IECs). In the U.s.a., Institutional Review Boards (IRBs) have oversight and must abide by the United States Food and Drug Administration (FDA) regulations. Countries other than those in the European Union and the United States take individual committees and regulations.
There are 2 types of IRBs and I ECs, local and central. Local comm ittees back up individual research institutions and are responsible for reviewing only their clinical trials. Cen tral boards oversee the review of clin ical studies for many different organizations.
More specific guidelines on IRB and IEC responsibilities, composition, functions, and operations are listed in this annex .
How are IRBs and IECs formed?
Per the FDA, each IRB/ I EC should include five or more members and come from various backgrounds . One of these representatives should come up from a non-scientific background and is referred to as a lay member. Another should be an independent member who is not affiliated with the clinical site or medical institution. All members should exist competent i ndividuals who are able to empathise all aspects of the clinical trial procedure.
The Food and Drug Assistants ( FDA ) has compiled a list of frequently asked question due south and their respective answers to provide clarity on United States regulatory procedures . This document explains how IRBs are created , outlines their respons ibilities, and g ives item on various questions one may encounter during the approval process.
A like directive has been provided for countries in the European Union. Once again , the arduous clinical trial process is cleaved down into more manage able data, but the reader will proceed to realize the numerous hurdles to be overcome.
Why are IECs and IRBs so critical?
Independent Ethics Committees and Institutional Review Boards are necessary to monitor all aspects of clinical trials, ensuring that man trial participants' condom is their top priority. They review each study through neutral optics, evaluating the risks and benefits throughout every clinical trial process step.
How long does it take to receive IRB/IEC approvals?
As explained earlier, each aspect of the clinical trial, including patient payment guidelines, must be approved by the IRB earlier any patient recruitment and enrollment are permitted to brainstorm. Generally, an IRB submission takes one month to receive approval, with expedited/exempt approvals returned in about two weeks. If there are whatever details of the report that do non align with IRB standards, the IRB refuses approving and suggests amendments to receive blessing. Just one minor change could upshot in weeks or months of delays for a clinical trial, leading to loss of money for the sponsor and loss of much-needed treatment for trial participants.
Each country beyond the earth has its regulations for stipend and per diem payments, patient reimbursements, and taxation reporting of income related to clinical trial participation. When conducting a trial at sites in various countries, understanding these unique requirements is hugely time-consuming and overwhelming. Ofttimes, countries will brand changes to their guidelines, causing the need to quickly pivot to accommodate to these updates and adhere to the new rules.
Equally your full-service provider of logistical support, Clincierge has the expertise needed to help assess the feasibility of supporting patients in specific regions worldwide. We streamline the process past providing required documents in multiple languages as well as manage any document revisions. Our squad keeps abreast of current regulations in each country and provides recommendations to ensure compliance and adherence to specific guidelines.
Clincierge has the expertise needed to help you navigate the complexities involved with IRBs and IECs across the globe. These services volition ease the burdens associated with your clinical trial regulatory processes. We handle:
- Information Compilation and Document Submissions
- IRB/EC Submissions
- Regulatory Reports
- Study Amendment Documentation
- Study Termination Documentation
Remove barriers to clinical trial participation and improve your trial's performance
Source: https://www.clincierge.com/irb-ec-critical-impact-on-clinical-trials/
Post a Comment for "Institutional Review Board/ethics Committee What Is the Role of"